Organization
Since 2024, Sanquin has operated under a new structure where the entire organization within the Sanquin Foundation – comprising 11 departments – reports to a single Executive Board (RvB). The previous divisions (Blood bank, Research & Lab Services, and Corporate Staff & Corporate Services) have been dissolved. The 11 departments now represent clusters of activities that integrate across the organization.
Organizational charts
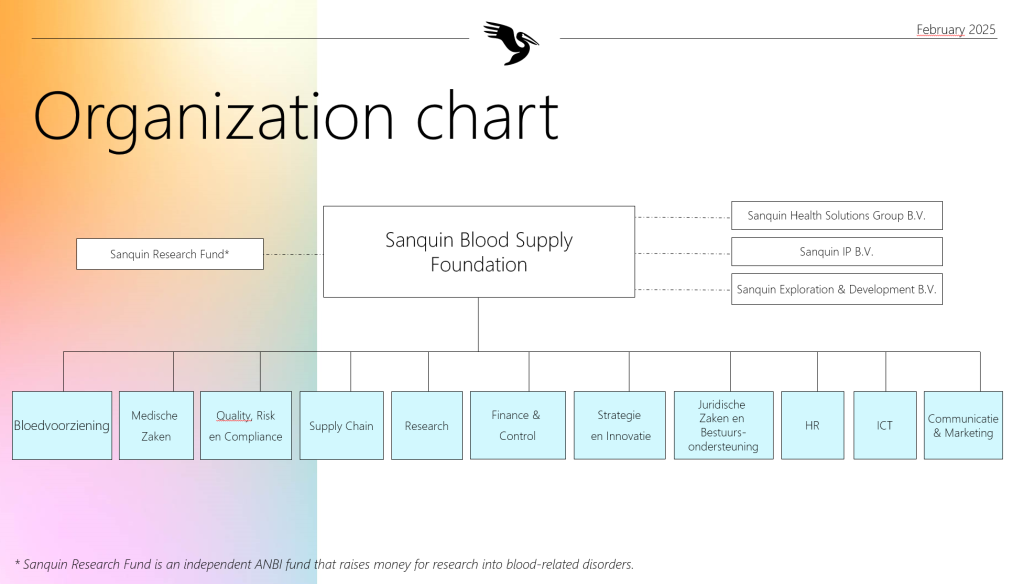
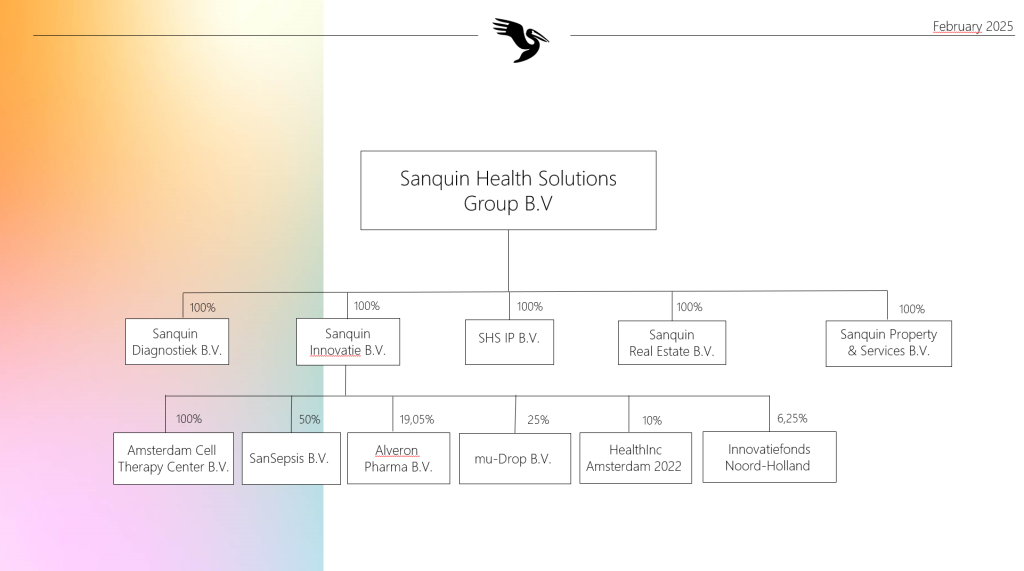
Advice and assessment
The following advisory councils and assessment committees play an active role within Sanquin:
- Donor councils and complaints committee
- Ethics Advisory Council
- Institutional Review Board
- Medical Advisory Council
- National User Council
- Scientific Advisory Board
Annual Reports
Our annual reports provide a retrospective view of the preceding year, detailing our activities and financial performance. Within these reports, you can find our achievements for patients, donors, and society as a Foundation, through the Sanquin Research Fund, and within Sanquin Health Solutions.
Annual report 2024 (in Dutch)
Annual report archive
For the annual reports of the last 5 years, see: Annual report archive.
ANBI Status
Stichting Sanquin (the part focused on public benefit) has been granted ANBI status. ANBI stands for Algemeen Nut Beogende Instelling, which translates to Public Benefit Organization. This status offers tax advantages for both the organization and Dutch taxpayers who donate, for example through gifts, to Sanquin.
An ANBI serves the public interest in various ways. It can be a charity, a cultural or scientific institution, or even a church or an organization with a specific philosophical basis. To maintain ANBI status, an organization must dedicate at least 90% of its efforts to the public good, must not aim to generate profit, and must adhere to integrity standards. Furthermore, an ANBI's capital is subject to limitations, and there are stringent regulations regarding expense reimbursements. Additionally, its costs and expenditures must maintain a reasonable balance.
Sanquin Blood Supply Foundation ANBI publications (Dutch):
- Stichting Sanquin Bloedvoorziening ANBI-publicatie 2023 (PDF, 456 kB)
- Stichting Sanquin Bloedvoorziening ANBI-publicatie 2022 (PDF, 259 kB)
- Stichting Sanquin Bloedvoorziening ANBI-publicatie 2021 (PDF, 259 kB)
- Stichting Sanquin Bloedvoorziening ANBI-publicatie 2020 (PDF, 260 kB)